  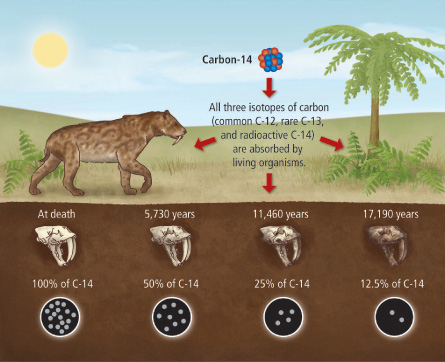
Since natural carbon contains all these isotopes mixed together, anything that takes up carbon from natural sources will have the same mix of isotopes in its carbon. It makes up about one part in a trillion of natural carbon. The type of carbon that we are interested in, because it is radioactive, is carbon with eight neutrons: carbon-14. There is also another stable, non-radioactive isotope of carbon with seven neutrons: carbon-13. About 99% of natural carbon is carbon-12. All carbon has six protons - that is what makes it carbon - and most carbon has six neutrons, making carbon-12 the normal isotope of carbon. We call them different isotopes, and name them by the sum of the number of protons and neutrons. All elements can have different numbers of neutrons in their nucleuses. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
